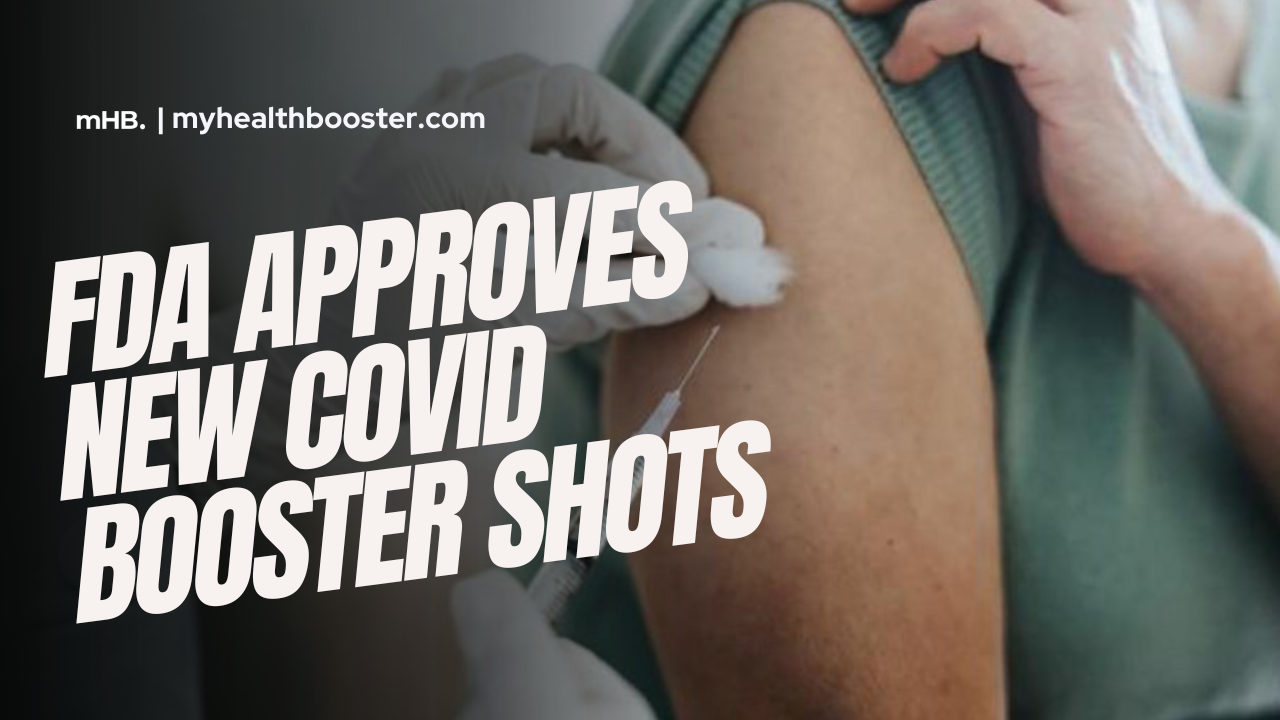
The U.S. Food and Drug Administration (FDA) has given the green light to updated COVID-19 booster shots from Pfizer and Moderna, marking a significant development in the ongoing battle against the virus. These booster shots are designed to combat the XBB.1.5 Omicron subvariant, are fully approved for individuals aged 12 and older, and are authorized for emergency use in those aged 6 months through 11 years.
Boosting Public Health Defenses
The approval of these booster shots, along with the recently approved RSV shots and the annual flu shot, forms a three-pronged public health strategy aimed at curbing the spread of multiple respiratory viruses during the upcoming winter season.
“Vaccination remains critical to public health and continued protection against serious consequences of COVID-19, including hospitalization and death,” emphasized Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research.
Who Can Receive the Booster Shots?
The FDA has outlined eligibility criteria for the updated COVID-19 booster shots:
- Age 5 and Older: Individuals aged 5 and older, regardless of previous vaccination, can receive a single dose of the updated COVID-19 vaccine, provided it’s been at least two months since their last COVID-19 vaccine dose.
- Ages 6 Months Through 4 Years: Those aged 6 months through 4 years who have been previously vaccinated against COVID-19 can receive one or two doses of the updated COVID-19 vaccine, depending on their previous vaccine history.
- Unvaccinated Ages: 6 Months Through 4 Years: Unvaccinated individuals aged 6 months through 4 years are eligible for three doses of the updated Pfizer COVID-19 vaccine or two doses of the updated Moderna COVID-19 vaccine.
The Role of the Booster Shots
Although the XBB.1.5 Omicron subvariant is no longer the dominant strain, closely related variants continue to circulate. Health experts believe that these booster shots will still offer valuable protection.
The Road Ahead
The Advisory Committee on Immunization Practices is scheduled to convene to make recommendations on who should receive the new booster shots. Following their guidance, Dr. Mandy Cohen, director of the U.S. Centers for Disease Control and Prevention, could provide final authorization, allowing vaccination efforts to commence.
Timing is Key
With COVID-19 cases on the rise again, the availability of this season’s COVID-19 vaccine is a timely development. The general population, aged 6 months and older, is now eligible to receive this year’s COVID-19 vaccine, even if they’ve never been vaccinated against COVID-19 before.
Cost Considerations
While most individuals with private and public health insurance should still receive these booster shots for free, there’s a cost attached for the uninsured. The federal government will not cover the expense of these shots. The expected price per dose is between $110 and $130.
The Biden administration has introduced a “bridge” program to offer uninsured individuals access to free booster shots through 2024, aiming to ensure that everyone has an opportunity to receive the necessary protection against COVID-19.
The availability and coverage of these shots may vary depending on individual insurance plans, and uninsured individuals are encouraged to explore options at community health centers.
SOURCES: U.S. Food and Drug Administration, news release, Sept. 11, 2023;
Pfizer Inc., news release, Sept. 11, 2023;
Moderna Inc., news release, Sept. 11, 2023;
Amesh Adalja, senior scholar, Center for Health Security, Johns Hopkins University, Baltimore; NBC News